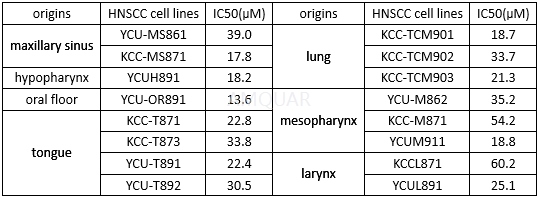
| NCT Number | Conditions | Interventions | Sponsor/Collaborators | Phases | Start Date | Last Updated |
| NCT01184482 | Colorectal Cancer|Lung Cancer|Head and Neck Cancer | Drug: cetuximab and lapatinib | Georgetown University|GlaxoSmithKline | Phase 1 | 2010-06-01 | 2013-10-29 |
| NCT02101905 | Anaplastic Astrocytoma|Anaplastic Ependymoma|Anaplastic Oligodendroglioma|Gliosarcoma|Mixed Glioma|Recurrent Adult Brain Neoplasm|Recurrent Glioblastoma | Other: Laboratory Biomarker Analysis|Drug: Lapatinib Ditosylate|Other: Pharmacological Study|Procedure: Therapeutic Conventional Surgery | National Cancer Institute (NCI) | | 2014-03-01 | 2017-03-10 |
| NCT01947023 | BRAF V600E Mutation Present|BRAF V600K Mutation Present|Metastatic Malignant Neoplasm in the Thyroid Gland|Recurrent Thyroid Gland Carcinoma|Unresectable Thyroid Gland Carcinoma | Drug: Dabrafenib|Other: Laboratory Biomarker Analysis|Drug: Lapatinib Ditosylate|Other: Pharmacological Study | National Cancer Institute (NCI) | Phase 1 | 2013-08-01 | 2016-12-21 |
| NCT00694252 | Metastatic Breast Cancer | Drug: Lapatinib | University Hospital of Crete | Phase 2 | 2008-07-01 | 2015-09-25 |
| NCT00975988 | Cancer | Drug: TYKERB庐 tablets | GlaxoSmithKline | | 2010-01-01 | 2014-10-23 |
| NCT00103194 | Recurrent Prostate Cancer|Stage I Prostate Cancer|Stage IIA Prostate Cancer|Stage IIB Prostate Cancer|Stage III Prostate Cancer | Drug: lapatinib ditosylate|Other: laboratory biomarker analysis | National Cancer Institute (NCI) | Phase 2 | 2005-09-01 | 2014-09-19 |
| NCT00206427 | Breast Cancer | Drug: GW572016|Drug: lapatinib | Baylor Breast Care Center|GlaxoSmithKline | Phase 2 | 2004-08-01 | 2013-07-26 |
| NCT00650910 | Metastatic ErbB2|Neoplasms, Breast|Breast Cancer | Drug: lapatinib|Drug: Digoxin | GlaxoSmithKline | Phase 1 | 2008-04-01 | 2012-05-31 |
| NCT01281163 | Estrogen Receptor Negative|Estrogen Receptor Positive|HER2/Neu Positive|Progesterone Receptor Negative|Progesterone Receptor Positive|Recurrent Breast Carcinoma|Stage IV Breast Cancer | Drug: Akt Inhibitor MK2206|Other: Laboratory Biomarker Analysis|Drug: Lapatinib Ditosylate|Other: Pharmacogenomic Study|Other: Pharmacological Study | National Cancer Institute (NCI) | Phase 1 | 2011-01-01 | 2015-08-26 |
| NCT00863122 | Vestibular Schwannoma|NF2|Neurofibromatosis 2|Acoustic Neuroma|Auditory Tumor | Drug: lapatinib | Sidney Kimmel Comprehensive Cancer Center|The Children's Tumor Foundation|GlaxoSmithKline|New York University|Ohio State University|House Research Institute|Washington University School of Medicine|Weill Medical College of Cornell University|Massachusetts General Hospital | Early Phase 1 | 2009-06-01 | 2013-08-05 |
| NCT00614978 | Metastatic Breast Cancer|Brain Metastases|HER2 Positive | Drug: lapatinib and temozolomide | Jules Bordet Institute|GlaxoSmithKline|Schering-Plough | Phase 1 | 2008-01-01 | 2012-09-18 |
| NCT01290354 | Cancer | Drug: Lapatinib|Drug: [11C] lapatinib | GlaxoSmithKline | Phase 1 | 2011-09-01 | 2013-08-01 |
| NCT00114283 | Recurrent Salivary Gland Cancer|Recurrent Squamous Cell Carcinoma of the Hypopharynx|Recurrent Squamous Cell Carcinoma of the Larynx|Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity|Recurrent Squamous Cell Carcinoma of the Nasopharynx|Recurrent Squamous Cell Carcinoma of the Oropharynx|Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity|Recurrent Verrucous Carcinoma of the Larynx|Recurrent Verrucous Carcinoma of the Oral Cavity|Salivary Gland Squamous Cell Carcinoma|Stage IV Squamous Cell Carcinoma of the Hypopharynx|Stage IV Squamous Cell Carcinoma of the Nasopharynx|Stage IVA Salivary Gland Cancer|Stage IVA Squamous Cell Carcinoma of the Larynx|Stage IVA Squamous Cell Carcinoma of the Lip and Oral Cavity|Stage IVA Squamous Cell Carcinoma of the Oropharynx|Stage IVA Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity|Stage IVA Verrucous Carcinoma of the Larynx|Stage IVA Verrucous Carcinoma of the Oral Cavity|Stage IVB Salivary Gland Cancer|Stage IVB Squamous Cell Carcinoma of the Larynx|Stage IVB Squamous Cell Carcinoma of the Lip and Oral Cavity|Stage IVB Squamous Cell Carcinoma of the Oropharynx|Stage IVB Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity|Stage IVB Verrucous Carcinoma of the Larynx|Stage IVB Verrucous Carcinoma of the Oral Cavity|Stage IVC Salivary Gland Cancer|Stage IVC Squamous Cell Carcinoma of the Larynx|Stage IVC Squamous Cell Carcinoma of the Lip and Oral Cavity|Stage IVC Squamous Cell Carcinoma of the Oropharynx|Stage IVC Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity|Stage IVC Verrucous Carcinoma of the Larynx|Stage IVC Verrucous Carcinoma of the Oral Cavity|Tongue Cancer | Drug: lapatinib ditosylate|Other: laboratory biomarker analysis | National Cancer Institute (NCI) | Phase 2 | 2005-03-01 | 2015-04-14 |
| NCT00496366 | Breast Cancer|Metastatic Breast Cancer|Advanced Breast Cancer|HER2/Neu-positive Breast Cancer | Drug: Capecitabine|Drug: Lapatinib | Rutgers, The State University of New Jersey|National Cancer Institute (NCI)|Rutgers Cancer Institute of New Jersey | Phase 2 | 2007-07-23 | 2017-03-20 |
| NCT00912275 | Metastatic Breast Cancer | Drug: lapatinib plus oral vinorelbine | National Taiwan University Hospital|GlaxoSmithKline | Phase 1|Phase 2 | 2009-05-01 | 2012-11-15 |
| NCT00225758 | Metastatic Breast Cancer | Drug: Lapatinib | Dartmouth-Hitchcock Medical Center|University of Colorado, Denver|North Shore University Hospital | Phase 2 | 2006-01-01 | 2015-01-12 |
| NCT01245205 | HER2-positive Breast Cancer|Male Breast Cancer|Recurrent Breast Cancer|Stage IIIB Breast Cancer|Stage IIIC Breast Cancer|Stage IV Breast Cancer|Unspecified Adult Solid Tumor, Protocol Specific | Drug: Akt inhibitor MK2206|Drug: lapatinib ditosylate|Other: pharmacological study|Other: laboratory biomarker analysis | National Cancer Institute (NCI) | Phase 1 | 2010-11-01 | 2014-10-22 |
| NCT00973739 | Neurofibromatosis 2|Vestibular Schwannoma | Drug: Lapatinib | New York University School of Medicine|GlaxoSmithKline | Phase 2 | 2009-09-01 | 2016-02-19 |
| NCT00470704 | Breast Cancer | Drug: Lapatinib|Drug: Herceptin | Nancy Lin, MD|GlaxoSmithKline|Dana-Farber Cancer Institute | Phase 2 | 2007-05-01 | 2017-01-03 |
| NCT00820924 | Neoplasms, Breast | Drug: LAPATINIB | GlaxoSmithKline | Phase 2 | 2008-06-01 | 2012-03-22 |
| NCT01283789 | Metastatic Breast Cancer | Drug: Lapatinib and RAD-001 | University of Kansas Medical Center|Novartis|GlaxoSmithKline | Phase 2 | 2011-02-01 | 2017-02-06 |