-
生物活性
Forskolin is a labdane diterpene that is produced by the Indian Coleus plant (Plectranthus barbatus). Forskolin is commonly used as a tool in biochemistry to raise levels of cyclic AMP (cAMP) in the study and research of cell physiology. Forskolin activates the enzyme adenylyl cyclase and increases intracellular levels of cAMP. cAMP is an important second messenger necessary for the proper biological response of cells to hormones and other extracellular signals. It is required for cell communication in the hypothalamus/pituitary gland axis and for the feedback control of hormones. Cyclic AMP acts by activating cAMP-sensitive pathways such as protein kinase A and Epac.Forskolin is an inhibitor of AChR, interacts directly with the catalytic subunit of A cyclase (adenylate cyclase), activating the enzyme and raising intracellular levels of cAMP. Forskolin is cell permeable and active in vivo producing positive inotropic, platelet anti-aggregatory and anti-hypertensive effects in vitro. Has also been shown to exhibit smooth muscle relaxant activity, decrease in intraocular pressure and the promotion of hormonal release from the pituitary glands in vitro. Inhibits MAP kinase, induces phosphorylation of CREB, connexin 43, Bad, ATF-1 and PKB.

Forskolin shows inhibition in the case of the isoproterenol (EC50 = 0.5μM forskolin) or direct Ns activation (EC50 = 0.5μM forskolin) stimulation of cyclic AMP production.[5]
-
体外研究
-
体内研究
-
激酶实验
Adenylate cyclase assay[6]
Adenylate cyclase activity was assayed by the conversion of ATP into cAMP in the presence of 1mM isobutylmethylxanthine and an ATP regenerating system as described by Scarpace and Abrass. Approximately 75μg of membrane protein plus stimulator (forskolin) was incubated for 15 min at 37°C in a total volume of 150μI. The reaction was terminated by addition of 700μI of cold 5.3mM CaCI2and immersion into a salt-ice bath. Assay tubes were centrifuged at 9000 g and the supernatant assayed for cAMP. cAMP was quantified by radioimmunoassay. Protein was assayed with bovine serum albumin as the standard.
Preparation of J774 cell membranes[5] In some experiments the cells were preincubated either for 24 h with 120ng/ml pertussis toxin or for 4 h with 500ng/ml cholera toxin before collection for membrane preparations. The cells in suspension were sedimented by centrifugation for 10 min at 900×g at 4°C. They were washed three times with ice-cold 17mM Tris : 110mM NaC1, pH 7.5, then suspended (5x 106cells/ml) in pre-cooled 5mM Tris : 5mM sodium β-glycerophosphate buffer, pH 7.5, containing 2mM EDTA, 1mM dithiothreitol, 1mM benzamidine, 0.04mM leupeptin, 80KIU/ml aprotinin, 1μg/ml soybean trypsin inhibitor and 0.1mM phenyl methylsulfonyl fluoride (lysis buffer). The cells were allowed to swell for 10 min then were broken by 80 strokes in a Dounce homogenizer at 4 ° C. The nuclei and cell debris were removed by centrifugation at 500 × g for 5 min. The supernatant was centrifuged for 30 min at 37000 x g. The pellet was resuspended in the same volume of lysis buffer and the suspension was centrifuged under the same conditions. The membranes were suspended to a concentration of 0.5-1mg protein/ml in 25mM Tris: 25mM β-glycerophosphate: 1mM EDTA buffer, pH 7.8, and stored under liquid nitrogen.
Adenylate cyclase assay
Adenylate cyclase activity was determined in 25mM Tris : 25mM β-glycerophosphate buffer (pH 7.5) containing 0.3mM EDTA, 5mM free MgCl2, 0.2mM ATP, 1μCi [α-32p]ATP, 10mM phosphocreatine, 10 U/ml creatine phosphokinase, 0.05mM Ro 20-1724, 0.5 U/ml adenosine deaminase. The effects of adenosine were suppressed under these conditions. We had established in preliminary experiments that the basal and the isoproterenol-stimulated adenylate cyclase activities were optimum with 0.2mM ATP, 5mM MgCl2and 10μM GTP. Various concentrations of GTP, GppNHp, MnCl2, isoproterenol, forskolin, forskolin derivatives or 10mM sodium fluoride were added. The reactions were initiated by the addition of membranes (6-20μg membrane protein per assay sample) in a final volume of 50μl. The reactions were terminated after 10 min at 30 °C by the addition of 1 N HC1 and 1μM cyclic AMP and boiling for 5 min. The mixture was neutralized with 3.34 M imidazole. [32p]Cyclic AMP was extracted on a column filled with aluminum oxide following elution with 10mM imidazole (pH 7.5) and was counted for radioactivity. The assays were performed in triplicate. Adenylate cyclase activity was expressed as the cyclic AMP (pmol/mg protein) formed in 10 min. The actual values for the basal or stimulated cyclic AMP levels varied from one experiment to another, depending on the membrane preparation used and on the day of the assay. Thus, the results shown in the curves are expressed as the values of one representative experiment from a series of 2-9 experiments. The relative effects of forskolin, either inhibitory or stimulatory, however, were within a constant range.
Protein determination
Protein was assayed with bovine serum albumin as the standard

-
细胞实验
Cell culture[7]
The U937 cell line was prepared. Cells were cultured in suspension at 37oC in RPMI 1640 medium supplemented with 300 mg/1L-glutamine, 10% fetal calf serum and 40μg/ml gentamicin in a 5% CO2humidified incubator. Cells were routinely passaged every 3–4 days and seeded at a density of 0.5x105cells/ml.
cAMP assay Cells were stimulated for 9 min with different concentrations of forskolin or histamine H2agonist (dimaprit) in Hanks’ solution with 1mM 3-isobutyl-1-methylxanthine (IBMX). The reaction was terminated by centrifugation for 3 min at 3000 x g, followed by the addition of 1 ml absolute ethanol. Supernatants were centrifuged for 10 min at 3000 x g, and the ethanol phase was dried and resuspended in 50mM Tris–HCl buffer. cAMP content was determined by means of competition with [3H]cAMP for protein kinase A. Pretreatment of cells (desensitization experiments) with forskolin was performed in Hanks at 37oC in a 5% CO2humidified atmosphere. Cells were exposed to 75μM forskolin (maximal response) for periods ranging from 1 min to 24 h in the absence of IBMX. Thereafter, cells were washed and resuspended in Hanks with 1 mM IBMX at a cell density of 106cells/ml and exposed for 9 min to 75μM forskolin or 10μM dimaprit, to determine whether the adenylyl cyclase could still generate cAMP. In every experiment, each point was assayed in triplicate.
-
动物实验
Animals and Treatment[8]
Male APP/PS1-21 mice were obtained. Heterozygous male APP/PS1-21 mice were bred with wild-type C57BL/6J females. Offspring were tail snipped and genotyped using PCR with primers specific for the APP sequence (Forward: “GAATTCCGACATGACTCAGG,” Reverse: “GTTCTGCTGCATCTTGGACA”). Mice were kept in a 12-hour dark/12-hour light environment and were given ad libitum access to water and food.
ForskolinTreatment
For oral treatment, forskolin (>98% pure) was suspended in 1% carboxymethylcellulose (CMC) and administered by gavage at a dose of 100 mg/kg. Control mice received an equivalent volume of CMC.
Mice were randomly assigned to groups (n=6/group, 3 males and 3 females for each group) at the age of 5 months and used for the experimental treatments: daily oral administration of forskolin (100 mg/kg bodyweight, suspended in CMC), or the same volume of 1% CMC by gavage. The experiment lasted 10 days and consisted of 3 groups: Forskolin treatment, vehicle control, and controls without any treatment.
Design and Evaluation of Nest Construction Assay
A nest construction assay was modified (22) to determine the deficits in affiliative/social behavior of APP/PS1 mice and potential changes following treatment. Mice were individually housed for at least 24 hours in clean plastic cages containing 1 cm of wood chip bedding. Mice were tested in counterbalanced groups of mixed genotypes and genders to reduce variability in housing conditions. Two hours prior to the onset of the dark phase, individual cages were supplied with paper towel torn into 5 x 5 cm squared pieces. The next morning, cages were inspected for nest construction. Paper towel nest construction was scored by a 3-point system: 1) no biting or tears on the paper; 2) moderate biting and/or tears on the paper but no coherent nest; 3)the vast majority of paper torn into pieces and grouped into a corner of the cage.
Forskolin-treated and control mice were killed after 10 days of treatment. Nest scores were given by 3 independent observers who were not aware of the treatment categories.
Social Interaction Assay
The apparatus was made up of a 3-chambered box. Prior to the experiment, 2 dividing walls were made with openings in order to allow free access into each chamber. Moreover, each chamber was cleaned and paper chip bedding was added for each single trial.
The test mouse (ie, transgenic mouse, treated or not) was placed in the middle chamber and allowed to accommodate for 5minutes. After the accommodation period, a stranger mouse (stranger 1, with which the test mouse was not familiar and had no previous contact) was placed in one of the side chambers. The stranger 1 was placed, respectively, in the left and in the right side chamber between trials. The mice serving as strangers were male C57BL/6J that had previously been accommodated in small cages. The entries to the side chambers were made free and the test mouse was allowed to explore the entire social test box for 10 minutes. We then measured the time spent in each chamber. After the first 10 minutes, each test mouse was submitted to a second 10 minutes session to quantitate social preference toward a new stranger mouse. Thereafter, another unfamiliar and unknown mouse (stranger 2) was introduced in the chamber that was kept empty during the first 10 minute. In this second phase, the test mouse had to choose between the stranger 1 (unfamiliar but with which it was already in contact) and the newly introduced stranger 2 mouse (also unfamiliar). Measures were taken to estimate the amount of time spent in each chamber of the equipment during the second 10-minute session.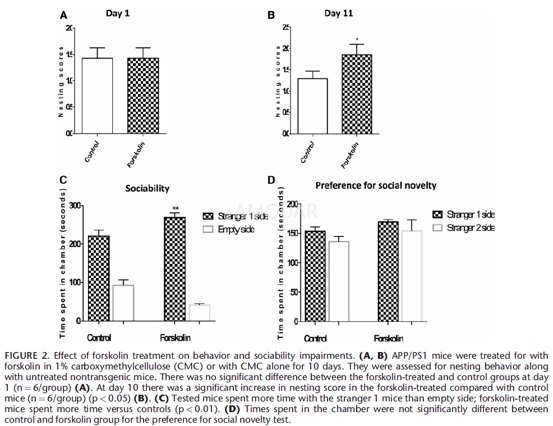
-
不同实验动物依据体表面积的等效剂量转换表(数据来源于FDA指南)
|  动物 A (mg/kg) = 动物 B (mg/kg)×动物 B的Km系数/动物 A的Km系数 |
|
例如,已知某工具药用于小鼠的剂量为88 mg/kg , 则用于大鼠的剂量换算方法:将88 mg/kg 乘以小鼠的Km系数(3),再除以大鼠的Km系数(6),得到该药物用于大鼠的等效剂量44 mg/kg。
-
参考文献
[1] Hartzell HC BD. Differences in effects of forskolin and an analog on calcium currents in cardiac myocytes suggest intra- and extracellular sites of action. Mol Pharmacol. 1992;41(5):880-888.
[2] Lal B GA, Rajagopalan R, Ghate AV. Hydroxyacyl derivatives of forskolin--their positive inotropic activity. Bioorg Med Chem. . 1998 6(11):2061-2073.
more
分子式
C22H34O7 |
分子量
410.51 |
CAS号
66575-29-9 |
储存方式
﹣20 ℃冷藏长期储存。冰袋运输 |
溶剂(常温)
|
DMSO
25 mM |
Water
<1 mg/mL |
Ethanol
10 mM |
体内溶解度
-
Clinical Trial Information ( data from http://clinicaltrials.gov )
| NCT Number | Conditions | Interventions | Sponsor/Collaborators | Phases | Start Date | Last Updated |
| NCT00864578 | Primary Open Angle Glaucoma|Intraocular Pressure | | Sooft Italia | | 2009-04-01 | 2012-06-05 |
| NCT00863811 | Primary Open Angle Glaucoma|Intraocular Pressure | | Sooft Italia | | 2009-04-01 | 2012-06-05 |
| NCT01254006 | Glaucoma | Drug: forskolin, rutin and vitamins B1 and B2 | University of Roma La Sapienza|vingolo enzo maria|domanico daniela|salvatore serena | | null | 2010-12-03 |
| NCT02143349 | Metabolic Syndrome|Obesity | Dietary Supplement: Coleus forskohlii extract|Other: Placebo | Olive Lifesciences Pvt Ltd|College of Health and Biomedicine, Victoria University | Phase 3 | 2014-04-01 | 2014-05-17 |
| NCT02807415 | Cystic Fibrosis | Drug: Lumacaftor plus Ivacaftor | Hannover Medical School|Heidelberg University|University of Giessen | | 2016-06-01 | 2016-06-22 |
| NCT02586883 | Idiopathic Dilation of the Bronchi | Other: bronchial ddp test | Assistance Publique - H么pitaux de Paris | | 2016-04-01 | 2016-12-02 |
注:以上所有数据均来自公开文献,并不保证对所有实验均有效,数据仅供参考。
-
相关化合物库
-
使用AMQUAR产品发表文献后请联系我们





















