-
生物活性
The IC50 of proliferation: 0.146 μM inHH cells; 2.062 μM in HuT78 cells; 2.697 μM in MJ cells; 1.375 μM in MylA;1.510 μM in SeAx cells.[2][17][18][19]
Antiproliferative activity human cell lines: 0.7μM of NB4; 3.4μM of H460; 1.2 μM of HCT-116.[2][17][18][19]
In vitro activity against P. falciparum is ~100-300nM; In vivo against Plasmodium parasites for P. falciparum and P. vivax is IC50 310 and 170 nM, respectively.[2][17][18][19]
Inhibition of recombinant PfHDAC1 enzymatic and P.falciparum 3D7 is IC50 59 nM and 47 nM, respectively.[2][17][18][19]
-
体外研究
The HDAC inhibitor vorinostat (SAHA) down-regulates C-FLIP and sensitizes human Non-small Cell Lung carcinoma cell lines to Ionising radiation.[1] Vorinostat induces cell death, cell cycle arrest and accumulation of acetylated histones in cutaneous T-cell lymphoma cells.[2] SAHA may suppress the aggressive phenotypes of glioma cells by increasing the expression of acetyl-histones 3 and 4, which were recruited to the promoters of p21, p27, Cyclin D1, c-myc and Nanog to down-regulate their transcriptional levels.[3][4] SAHA inhibits cervical cancer cell proliferation by and blocking the cell cycle at G1/S transition inhibiting the transcription initiation of HPV18 E6 and E7 genes.[5] SAHA induced hyperacetylation of histone H3 K9, K14 and K23 and triggered apoptosis. Besides, SAHA also caused cell cycle arrest by upregulation of CDKN1A and downregulation of CCND1 encoding p21WAF1/CIP1 and cyclin D1 proteins, respectively.[6] Vorinostat exerts a positive effect on neuritogenesis via activation of MEK1/2 &PI3K pathways involving an upstream kinase, TrkA.[7] Vorinostat is effective in reducing survival (via apoptosis activation) and ACTH secretion (via NFkB overexpression) in AtT20 and human tumors.[8] Combined treatments with vorinostat promote the cytotoxicity of cisplatin and induce the expression of vorinostat-regulated acetyl proteins, eventually enhancing antitumor effects in SCLC cell lines.[9] vorinostat,s differential modulation of 3D nuclear architecture in normal and abnormal cells could play a functional role in its anti-cancer action.[10] SAHA induced MET activation, which in turn contributed to the resistance to SAHA in solid cancer cells.[11] Vorinostat/SAHA-induced apoptosis in malignant mesothelioma is FLIP/caspase 8-dependent and HR23B-independent.[12] SAHA Suppresses IL-1β-Induced Matrix Metallopeptidase-13 Expression by Inhibiting IL-6 in Osteoarthritis Chondrocyte. [13] SAHA causes bone loss in vivo by reducing osteoblast number, even while increasing activity of pre-existing mature osteoblasts.[14] Histone deacetylase inhibitor SAHA mediates mast cell death and epigenetic silencing of constitutively active D816V KIT in systemic mastocytosis.[15] Vorinostat treatment limits the aberrant up-regulation of cholesterol biosynthesis and uptake that arises from lysosomal sequestration of cholesterol in NP-C disease.[16]
-
体内研究
2% DMSO, 40% PEG 300, 5% Propylene glycol, 1% Tween 80
-
激酶实验
HDAC enzyme activity assay[20]
The Color-de-Lys system (Colorimetric Histone de Acetylase Lysyl Substrate/Developer) is a sensitive and convenient alternative to protocols utilizing radiolabeled, acetylated histones or peptide/HPLC methods for theassay of HDACs. Color-de-Lys- HDAC colorimetric activity assay kit (Enzo Life Sciences) was used according to manufacturer’s instructions to detect the enzymatic activity of HDACs present in the nuclear extracts isolated from HepG2 and SK-HEP-1 cells. This assay procedure has two steps; first, the Color de Lys Substrate that comprises an acetylated side chain of lysine is incubated with nuclear extract containing HDAC activity followedby test samples (SAHA or its analogs). Then the deacetylation process sensitizes the substrate and in the second step, the Color de Lys Developer that added to the substrate causes an increase in yellow color that is absorbed at 405 nm.
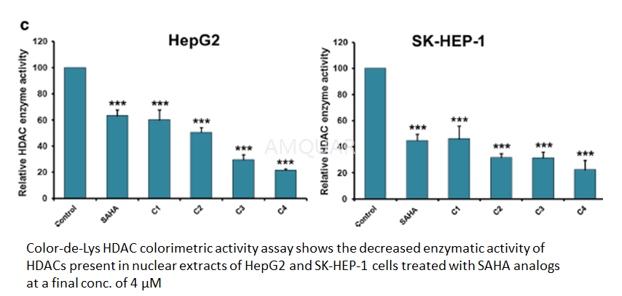
Histone deacetylase activity assay. [17]
For each HDAC assay, an aliquot corresponding to 20,000-40,000 dpm of histone H4 labeled with 3H-acetate Na salt (NEN no. NET003H) supplied in ethanol solution (1 mCi/ml), was added to each tube containing HDAC immunoprecipitated from 200 µg of total HeLa cell lysates resuspended in 25 µL of assay buffer (100 mM Tris-HCl pH 8, 2mM EDTA). Drugs at different concentrations were added and incubated for 90 minutes at 30° C. The reactions were terminated by adding 20 µl of stop buffer (0.5 N HCl, 0.08 M AcOH). 0.8 mL of TBME were added to extract released tritiated acetate. After centrifugation at 8,000 × g for 5 min, 600 µL of organic phase (upper phase) were transferred into a scintillation vial containing 3 ml scintillation liquid. The 50% inhibitory concentrations (IC50) for molecules were evaluated by the “ALLFIT” computer program by analyzing dose-response inhibition curves.
-
细胞实验
Cell culture
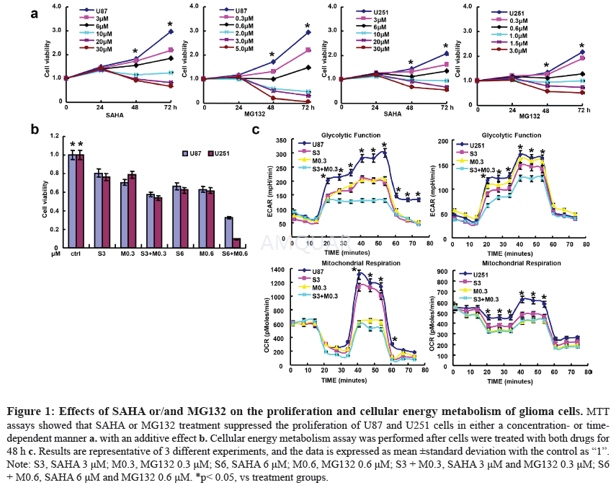
Glioma cells U87 and U251 were obtained from ATCC (Manassas, VA, USA) and grown in a humidified atmosphere of 5% CO2 at 37 °C in MEM (U87) or DMEM (U251) with 10% fetal bovine serum (FBS), 100 U/ml penicillin and 100 μg/ml streptomycin added. All cells were collected by centrifugation, washed in phosphate buffered saline (PBS) and subjected to RNA and protein extraction. We treated glioma cells to SAHA (Sigma, USA) and MG132 (Sigma, USA) for the following experiments.
Proliferation assay
Cells were planted at 2.0×103 cells per well in 96- well plates and maintained in media containing 10% FBS. At the time points, 20 μl of 5 mg/ml MTT was added to each well, and then incubated for 4 h at 37 °C. After that, the media was discarded, and 150 μl of DMSO was added to each well to dissolve the precipitates. The absorbance was measured at 490 nm using a microplate reader (M200pro, Switzerland).
Cellular energy metabolism
Mitochondrial respiration and glycolysis were measured by Seahorse XF Extracellular Flux Analyzer. Respiration and glycolysis are respectively measured as the rate of oxygen consumption (OCR) and extracellular acidification (ECAR). According to the standard protocol, we injected the regents in the following order: 10 mM glucose, 1.5 μM oligomycin (which blocks ATP synthase to assess respiration required for ATP turnover), 50 mM 2-DG for glycolysis assay and 1.5 μM oligomycin, 2 μM FCCP (a proton ionophore which induces chemical uncoupling and maximal respiration), 0.5 μM rotenone and antimycin A (which completely inhibits electron transport to measure non-mitochondrial respiration) for mitochondrial respiration. OCR and ECAR values were determined from 3 wells per sample and experiments were replicated.[3]
-
动物实验
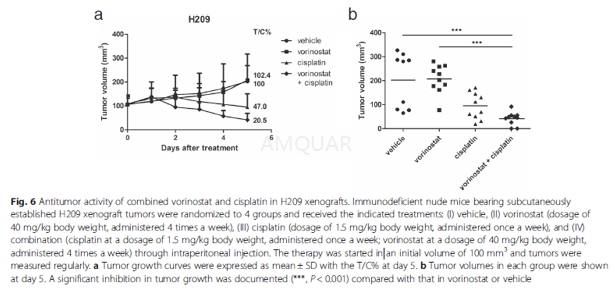
6-week-old male athymic nude mice (BALB/cAnN.Cg-Foxn1nu/ CrlNarl) were group housed (five mice per cage) under the condition of a 12-h light/dark cycle at 21–23 °C and 50–60 % humidity.
For implantation, one million H209 cells were harvested during the log-phase growth and resuspended in IMDM medium with Matrigel (BD, Franklin Lakes, NJ, USA) at a ratio of 1:1. Cells were then subcutaneously injected into the flanks of the mice. Once the tumor volume reached 100 mm3, the mice were randomized into four groups and administered the following agents through intraperitoneal injections: (I) vehicle, (II) vorinostat (at a dosage of 40 mg/kg body weight administered 4 times a week; vorinostat was first dissolved in DMSO and then added to a deionized solution with 5 % (v/v) polyethylene glycol 400 (PEG 400) and 5 % (v/v) Tween-80, and the final concentration of DMSO was 10 %). Tumor growth was monitored daily, and tumor volume (mm3) was defined as (l x w2)/2, where l is the length and w is the width (mm) of the tumor. The treatment was continued for 5 days, and the mice were euthanized 4 h after the final dose. According to the US National Cancer Institute protocols, tumor growth inhibition (T/C%) was calculated using the formula [(average volume of a treated group)/(average volume of a control group)] × 100 %; T/C% equal to or less than 42 % is considered significant antitumor activity.[9]
-
不同实验动物依据体表面积的等效剂量转换表(数据来源于FDA指南)
|  动物 A (mg/kg) = 动物 B (mg/kg)×动物 B的Km系数/动物 A的Km系数 |
|
例如,已知某工具药用于小鼠的剂量为88 mg/kg , 则用于大鼠的剂量换算方法:将88 mg/kg 乘以小鼠的Km系数(3),再除以大鼠的Km系数(6),得到该药物用于大鼠的等效剂量44 mg/kg。
-
参考文献
[1] McLaughlin KAJ, Stasik I, Prise KM, Johnston PG, Longley DB. 1132 The HDAC Inhibitor Vorinostat (SAHA) Down-regulates C-FLIP and Sensitizes Human Non-small Cell Lung Carcinoma Cell Lines to Ionising Radiation. European Journal of Cancer. 2012;48:S272-S273.
[2] Wozniak MB, Villuendas R, Bischoff JR, et al. Vorinostat interferes with the signaling transduction pathway of T-cell receptor and synergizes with phosphoinositide-3 kinase inhibitors in cutaneous T-cell lymphoma. Haematologica. 2010;95(4):613-621.
more
分子式
C14H20N2O3 |
分子量
264.3 |
CAS号
149647-78-9 |
储存方式
﹣20 ℃冷藏长期储存。冰袋运输 |
溶剂(常温)
|
DMSO
53 mg/mL at 25 ℃ |
Water
1.17 mg/mL at 25 ℃ |
Ethanol
3 mg/mL at 25 ℃ |
体内溶解度
5 mg/mL
-
Clinical Trial Information ( data from http://clinicaltrials.gov )
注:以上所有数据均来自公开文献,并不保证对所有实验均有效,数据仅供参考。
-
相关化合物库
-
使用AMQUAR产品发表文献后请联系我们