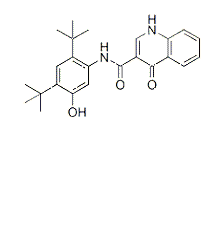
| NCT Number | Conditions | Interventions | Sponsor/Collaborators | Phases | Start Date | Last Updated |
| NCT02194881 | Cystic Fibrosis | Drug: CF patients with a G551D mutation and treated with Ivacaftor | Assistance Publique - H么pitaux de Paris|Vaincre la Mucoviscidose | | 2014-10-01 | 2016-07-31 |
| NCT01784419 | Cystic Fibrosis | Drug: ivacaftor | University of California, San Francisco | | 2013-10-01 | 2014-10-17 |
| NCT01705145 | Cystic Fibrosis | Drug: Ivacaftor | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 3 | 2013-01-01 | 2016-03-09 |
| NCT03085485 | Chronic Obstructive Pulmonary Disease|Chronic Bronchitis | Drug: Ivacaftor 150 MG|Drug: Placebo | University of Alabama at Birmingham|National Heart, Lung, and Blood Institute (NHLBI)|Vertex Pharmaceuticals Incorporated | Phase 2 | 2017-03-01 | 2017-03-14 |
| NCT02310789 | Cystic Fibrosis | Drug: Ivacaftor | Richard Barry Moss|Stanford University | Early Phase 1 | 2015-07-01 | 2016-04-23 |
| NCT01614457 | Cystic Fibrosis | Drug: Ivacaftor|Drug: Placebo | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 3 | 2012-07-01 | 2015-01-31 |
| NCT01946412 | Cystic Fibrosis | Drug: Ivacaftor | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 3 | 2013-12-01 | 2016-12-06 |
| NCT01707290 | Cystic Fibrosis | Drug: ivacaftor | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 3 | 2013-02-01 | 2016-05-10 |
| NCT02934698 | Cystic Fibrosis | Drug: Ivacaftor | Medical University of South Carolina|Vertex Pharmaceuticals Incorporated | Phase 3 | 2016-10-01 | 2016-10-14 |
| NCT02725567 | Cystic Fibrosis | Drug: ivacaftor | Vertex Pharmaceuticals Incorporated | Phase 3 | 2016-03-01 | 2017-03-22 |
| NCT01899105 | Cystic Fibrosis | Drug: lumacaftor|Drug: ivacaftor | Vertex Pharmaceuticals Incorporated | Phase 1 | 2013-07-01 | 2014-04-02 |
| NCT02412111 | Cystic Fibrosis | Drug: ivacaftor|Drug: VX-661|Drug: Placebo | Vertex Pharmaceuticals Incorporated | Phase 3 | 2015-06-01 | 2017-02-23 |
| NCT02135432 | Chronic Obstructive Pulmonary Disease | Drug: Ivacaftor|Drug: Placebo | University of Alabama at Birmingham | Early Phase 1 | 2015-05-01 | 2017-01-05 |
| NCT01897233 | Cystic Fibrosis | Drug: Lumacaftor|Drug: Ivacaftor | Vertex Pharmaceuticals Incorporated | Phase 3 | 2013-07-01 | 2016-10-07 |
| NCT01614470 | Cystic Fibrosis | Drug: Ivacaftor|Drug: Placebo | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 3 | 2012-07-01 | 2014-10-23 |
| NCT02565914 | Cystic Fibrosis | Drug: VX-661/ivacaftor|Drug: ivacaftor | Vertex Pharmaceuticals Incorporated | Phase 3 | 2015-08-01 | 2016-04-27 |
| NCT01685801 | Cystic Fibrosis | Drug: Ivacaftor|Drug: Placebo-matched-to-ivacaftor tablet | Vertex Pharmaceuticals Incorporated | Phase 2 | 2012-09-01 | 2015-04-30 |
| NCT00953706 | Cystic Fibrosis | Drug: Ivacaftor|Drug: Placebo | Vertex Pharmaceuticals Incorporated|Cystic Fibrosis Foundation Therapeutics | Phase 2 | 2009-09-01 | 2015-08-27 |
| NCT03068312 | Cystic Fibrosis | Drug: Ivacaftor|Drug: Placebo | Vertex Pharmaceuticals Incorporated | Phase 3 | 2017-04-25 | 2017-02-24 |
| NCT02742519 | Cystic Fibrosis | Drug: ivacaftor|Drug: Placebo | Vertex Pharmaceuticals Incorporated | Phase 3 | 2016-05-01 | 2016-12-12 |