细胞实验
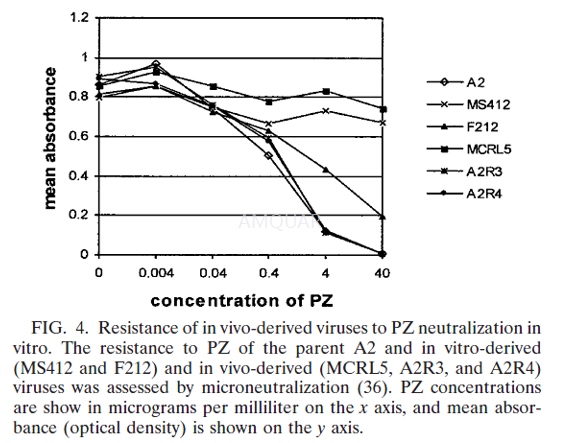
Microneutralization assay[1]
1,000 PFU of the standard viruses A2 andMS412, 1,000 PFU of the recovered viruses MCRL5 (rat
A-4), A2R3 (rat C-3), and A2R4 (rat C-4),or 5,000 PFU of standard virus F212 was incubated with serially diluted palivizumab(PZ) in the presence of guinea pig complement for 1 h at 37°C. The amount ofF212 was increased in response to reduced F212 growth in cell culture and toensure that the absorbance of the F212 wells would be similar to that of theother viruses in the final step of the assay. These neutralized viral solutionswere used to inoculate HEp2 cells in triplicate in96-well plates for 1 h at37°C. After 3 days, the cells were fixed with acetone. The amount of viralantigen present, measured by using a G-protein-specific enzymelinked immunosorbentassay, was used as a measure of viral replication. Mean absorbance at 450 nmwas recorded for triplicate wells.
动物实验
Animal model[1]
Cotton rats (Sigmodon hispidus) weretreated with intraperitoneal injections of cyclophosphamide (CY): 50 mg/kgthree times per week for at least 3 weeks prior to viral challenge. CY treatmentwas continued until the end of study. This led to marked leukopenia, with >80%reduction in total white blood cell counts compared to animals that did notreceive CY.
Experimentaldesign
Twelve cotton rats were divided into 4groups: A, B, C, and D (Fig. 1). Group D cotton rats (2 rats) did not receivepalivizumab (PZ) or CY and served as a control for white blood cell counts. Allother rats received CY. PZ was administered intramuscularly (i.m.) at 15 mg/kg.Group A (4 rats) received PZ 3 weeks post-CY treatment. Twenty-four hourslater, cotton rats in groups A, B, and C were infected intranasally with 5 x 105 PFU (50 μl) of A2 virus. Infections were performed under ketamine-xylazineanesthesia. Groups A and B (2 rats) received PZ 6 weeks after the initialinfections. Group C (4 rats) was treated with CY but did not receive PZ andserved as a negative (no PZ) control for prolonged viral replication. Nasalwash samples were obtained under anesthesia weekly (except week 10postinfection) from rats in groups A and B and inoculated onto HEp-2 cells forviral isolation. Infected HEp-2 cells provided RNA for reverse transcription(RT)-PCR and nucleotide sequence determination. Twelve weeks after the initialviral challenge, the experiment was terminated and the animals were sacrificed;lungs were harvested for RNA extraction and viral isolation.
Nasalwash and viral isolation
HEp-2 cells were split into six-well plates1 day prior to sampling. Each animal’s nose was washed with 25μlof transportation medium (Eagle minimum essential medium supplemented with 2%fetal bovine serum and antibiotics) under light ketamine-xylazine anesthesia.Nasal wash samples in 1 ml of transportation medium were kept on ice. Aftervortexing for30 s, 0.5 ml of medium was inoculated onto HEp-2 cells andabsorbed for 2 h before being replaced with fresh 10% fetal bovine serum–Eagleminimum essential medium. Cultures were maintained in the absence of PZ untilviral cytopathic effect (CPE) was evident or for at least 5 days. A secondpassage was conducted if no CPE was noted in passage one. Nasal wash sampleswith no CPE in the second passage were designated culture negative.
Cottonrat lung harvest and viral isolation
Cotton rats were sacrificed at 12 weekspost-viral infection, and their lungs were harvested for viral isolation and viralRNA extraction. Ground lung suspension (300 μl) wasabsorbed on HEp-2 cells for 2 h, washed twice, replaced with fresh medium, andmaintained for 4 days in the absence of PZ. Little or no CPE was seen after theinitial inoculation, and the medium was passed again onto fresh HEp-2 cells.Lung samples with no CPE shown in passage 2 were considered culture negative.
Proportion of viruses in the mixedpopulation in the lungs
The 603-bp RT-PCR products from cotton ratlung samples or HEp-2 cells infected with parental A2 virus were gel purifiedand cloned into pCR2.1-TOPO by following the manufacturer’s instructions.Between 20 and 50 colonies were picked from each cloned RT-PCR, and thenucleotide sequences of the insert cDNAs were determined. The proportion ofdiffering genotypes in each population was calculated based on the total numberof clones sequenced.



















